The European Rare Disease Registry Infrastructure (ERDRI) renders rare disease registries' data searchable and findable. This is achieved through the provision of following components: European Directory of Registries (ERDRI.dor), Central Metadata Repository (ERDRI.mdr), Pseudonymisation Tool (ERDRI.spider), Search broker (ERDRI.sebro).
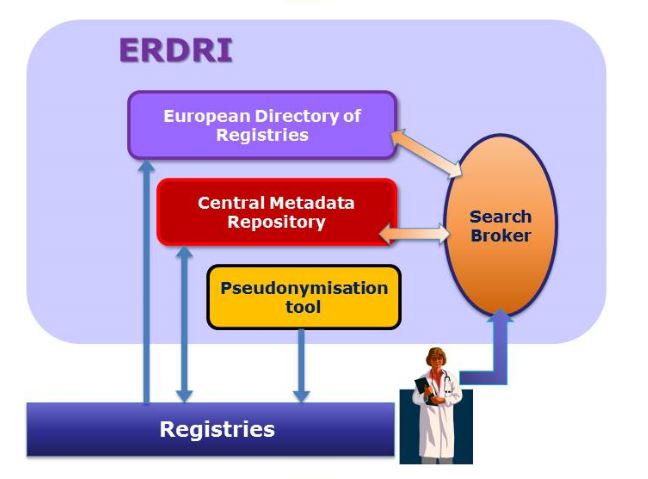
ERDRI supports existing registries in view of their interoperability and the creation of new registries.
ERDRI User access guide pdf
European Directory of Registries (ERDRI.dor)
ERDRI.dor provides an overview of the participating registries with their main characteristics and description.
Data input is performed by registry owners.
ERDRI.dor consists of eight sections with 42 data fields related to a registry of which 20 are obligatory.
ERDRI.dor user guide pdf
Central Metadata Repository (ERDRI.mdr)
ERDRI.mdr ensures semantic interoperability between RD registries. It stores all data elements (metadata) used by the participating registries, including the names of the data elements (designations) and their definitions.
Within ERDRI.mdr metadata items from any registry can be either uploaded automatically or inserted manually.
In case of establishing a new registry or amending an existing registry, a user can select from the metadata contained in ERDRI.mdr.
ERDRI.mdr user guide pdf
Pseudonymisation Tool (ERDRI.spider)
Service offering registries at local level the solution for patient pseudonymisation.
ERDRI.spider user guide pdf
Search broker (ERDRI.sebro)
ERDRI.sebro allows any user to retrieve metadata of interest within the ERDRI participating registries via ERDRI.sebro's connection to ERDRI.mdr and ERDRI.dor.